Periodic Table of Elements Project
Objective: Each student will randomly draw an element from the Periodic Table and become an expert on the element. You will present your research in a creative form of your choice and present your final project to the class.
Are you ready to have fun?
Are you ready to have fun?
|
Upcoming Deadlines
The Graphic Organizer & Final Element Project will be due on Friday, February 17, 2017 All students will give 1-3 minute presentation on the their project and what was learned. |
Weblinks
(Select one of the project below)
|
Periodic Table
|
Pictures of the Elements
|
Chemical Elements
|
Check out these Project Ideas!
Notes & Models
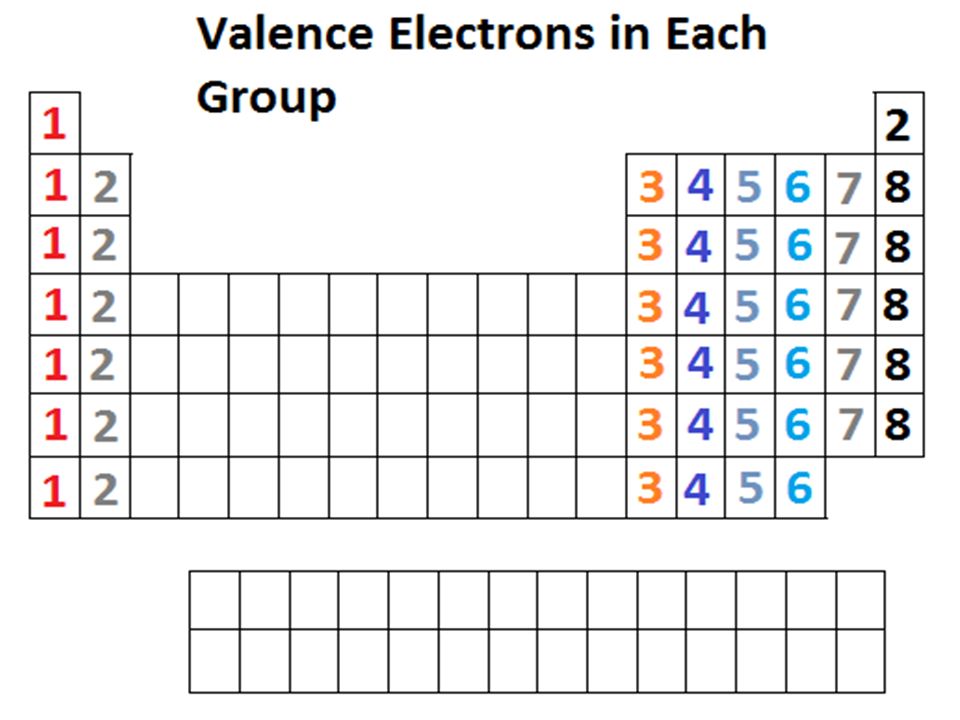
Valence Electrons |
Electronegativity |
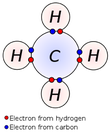
In chemistry, a valence electron is an outer shell electron that is associated with an atom, and that can participate in the formation of a chemical bond if the outer shell is not closed; in a single covalent bond, both atoms in the bond contribute one valence electron in order to form a shared pair. The presence of valence electrons can determine the element's chemical properties, such as its valence—whether it may bond with other elements and, if so, how readily and with how many. For a main group element, a valence electron can exist only in the outermost electron shell; in a transition metal, a valence electron can also be in an inner shell...Read More
|

Electronegativity, symbol χ, is a chemical property that describes the tendency of an atom to attract a shared pair of electrons (or electron density) towards itself.[1] An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity number, the more an element or compound attracts electrons towards it.
The term "electronegativity" was introduced by Jöns Jacob Berzelius in 1811,[2] though the concept was known even before that and was studied by many chemists including Avogadro.[2] In spite of its long history, an accurate scale of electronegativity was...Read More Online Research: Electronegativity Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. The Pauling scale is the most commonly used. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to Cesium and Francium which are the least electronegative at 0.7. |